电动病床注册技术审查指导原则(2017年修订版)
INFO
This content has been machine-translated from the Chinese original.
Official Full Text
Guidance on Technical Review for Electric Hospital Beds (Revised 2017)
This guidance document aims to standardize and clarify the technical review requirements for electric hospital bed products, and to provide specific guidance to applicants submitting applications for electric hospital bed products.
This guidance document does not include administrative approval requirements and is not legally binding. It should be used in accordance with relevant regulations.
This guidance document outlines general requirements for electric hospital bed products. Applicants should supplement and refine the content of their registration application documents based on the specific characteristics of their products. Applicants should also determine whether the specific content is applicable based on the characteristics of their products. If not applicable, they must provide a detailed explanation and supporting scientific evidence.
This guidance document was developed based on current regulations and standards, as well as the current level of knowledge. It also references relevant international standards, foreign regulations, and technical guidance documents. As regulations, standards, and scientific and technological development continue to evolve, the content of this guidance document will be adjusted accordingly.
I. Scope
These guidelines apply to electric hospital beds. Classified as Class II medical devices according to the "Medical Device Classification Directory" with the code number 6856.
II. Technical Review Points
(I) Product Name Requirements
The product name should typically be "electric hospital bed," or alternatively, the name specified in the industry standard YY 0571-2013 "Electrical Equipment for Medical Use - Part 2: Special Safety Requirements for Hospital Beds." The product name should comply with relevant regulations such as the "General Rules for Naming of Medical Devices" (State Food and Drug Administration Decree No. 19).
(II) Product Structure and Composition
An electric hospital bed consists of a bed surface, a bed frame, a drive mechanism, a control mechanism, and accessories.
The bed surface, bed frame, drive mechanism, control mechanism, and accessories should be determined based on the specific requirements of each product manufactured by different manufacturers. Generally, the bed surface consists of multiple support plates with different functions, such as a backrest, seat, thigh support, and calf support; the bed frame consists of a bed frame, headboard component, footboard component, left and right side rails, and casters; the drive mechanism consists of an electric actuator and a rechargeable battery. The electric actuator consists of a drive motor, reduction gear, screw, nut, guide sleeve, actuator, sliding seat, spring, housing, turbine, vane, and a micro-control switch; the control mechanism consists of a controller, control handle (wired/wireless), control panel, and CPR (backrest lowering mechanism) control buttons; and the accessories include: infusion stand, shoe rack, lifting rod, and urine bag hook.
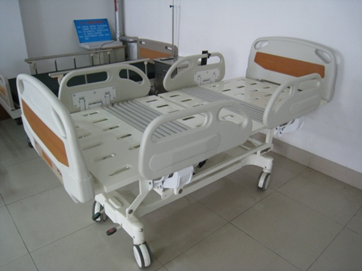
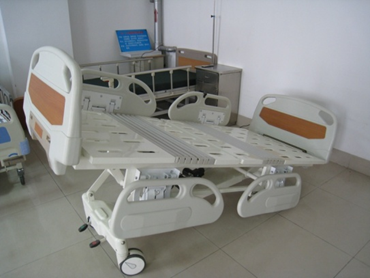
Figure 1 shows a schematic diagram of the patient bed's possible positions, and Figure 2 provides an example of a specific product and its components from a particular manufacturer for reference by reviewers.

1-1 Hospital Bed
1-2 full bed lift
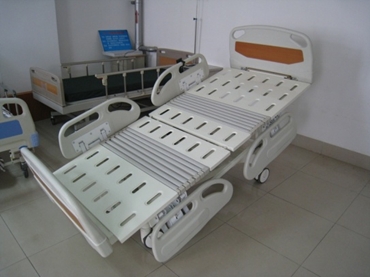
1-3 Bed tilting
1-4 Bed back tilt
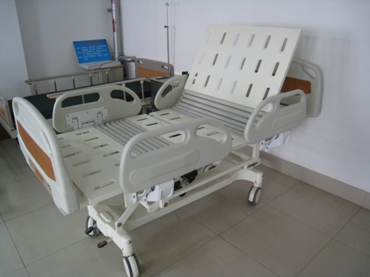
1-5 Back elevation

1-6: Elevation of lower limbs and back
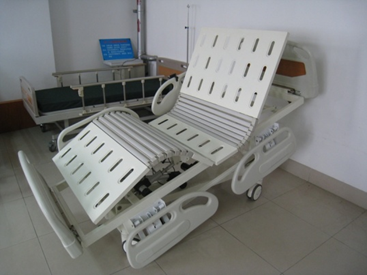
1-7 Overall forward tilt

1-8 Overall backward tilt

1-9 Bed
Figure 1 Schematic diagram of body position for various actions of an electric bed

Figure 2 Schematic diagram of an electric bed
(iii) Product Working Principle/Mechanism of Action
Product Working Principle: The applicant should provide a detailed explanation of the product's working principle, including a diagram of the working principle, and elaborate on how the various actions of the electric bed are achieved in conjunction with the diagram.
Since this product is a non-therapeutic medical device, this guidance does not include the product's mechanism of action.
(iv) Principles and Examples of Product Registration Unit Division
In principle, a standard electric bed can be divided into a single registration unit. If the principle, structure, or intended use undergoes a significant change, it should be divided into different registration units.
(v) Relevant Standards Applicable to the Product
The following are examples of commonly used standards related to electric beds:
Table 1. Relevant Product Standards
| Standard No. | Standard Name |
|---|---|
| GB/T 191-2008 | "Marking Requirements for Packaging and Storage" |
| GB 9706.1-2007 | "Medical Electrical Equipment Part 1: General Safety Requirements" |
| GB/T 14710-2009 | "Environmental Requirements and Test Methods for Medical Electrical Equipment" |
| GB/T 16886.1-2011 | "Biological Evaluation of Medical Devices Part 1: Evaluation and Testing in the Risk Management Process" |
| GB/T 16886.5-2003 | "Biological Evaluation of Medical Devices Part 5: In Vitro Cytotoxicity Test" |
| GB/T 16886.10-2005 | "Biological Evaluation of Medical Devices Part 10: Irritation and Delayed Hypersensitivity Test" |
| YY/T 0316-2008 | "Application of Risk Management to Medical Devices" |
| YY/T 0466.1-2009 | "Part 1: General Requirements for Symbols Used on Medical Devices, Labels, and Information Provision" |
| YY 0505-2012 | "Medical Electrical Equipment Part 1-2: General Safety Requirements - Parallel Standard: Electromagnetic Compatibility - Requirements and Test Methods" |
| YY/T 0708-2009 | Medical Electrical Equipment Part 1-4: General Safety Requirements: Programmable Medical Electrical Systems |
| YY 0571-2013 | "Part 2: Specific Safety Requirements for Electric Hospital Beds" |
Note: The standards listed above apply to the latest versions.
The above standards include those frequently referenced in the technical requirements and other relevant documentation for product registration. Some applicants may also refer to industry-specific standards and other specialized standards based on the characteristics of their products.
(6) Product Indications/Intended Use, Contraindications
Indications: For use in adult patients under medical supervision for diagnosis, treatment, or monitoring, to support the patient's body and achieve the required clinical position.
Intended Use Environment: Medical institution wards (patient rooms, observation rooms, etc.).
Target Population: Adult patients.
Contraindications: No relevant reports have been found.
(7) Principal Risks of the Product
The risk management activities should span the entire lifecycle of the product, from design and production to post-market use and disposal. The risk management plan of the applicant should be complete, especially the risk analysis and evaluation process for post-market management. For unknown risks in pre-market risk management, information should be collected after launch, and risks should be evaluated and controlled promptly upon discovery, with the risk management documents updated.
The risk management report for the electric bed should comply with the requirements of YY/T 0316-2008 "Application of Risk Management to Medical Devices," including identifying hazards related to the product, estimating and evaluating associated risks, controlling these risks, and monitoring the effectiveness of the controls.
The main review points include:
The determination of safety characteristics related to the product can refer to Appendix C of YY/T 0316-2008 "Application of Risk Management to Medical Devices";
The determination of hazards, potential event sequences, and hazard situations can refer to Appendix E and I of YY/T 0316-2008 "Application of Risk Management to Medical Devices";
The risk control plan and implementation, as well as the methods for comprehensively evaluating the acceptability of residual risks and monitoring production and post-production, can be referenced from Appendix F, G, and J of YY/T 0316-2008 "Application of Risk Management for Medical Devices."
Main Hazards of the Product
4.1 Energy Hazards
Electromagnetic energy: This includes the impact of fluctuations in the power supply on the device, leakage currents, and electromagnetic interference caused by equipment commonly used with the electric bed on the bed, as well as the impact of the electromagnetic field generated by the electric bed on equipment commonly used with it.
Thermal Hazards: Including burns caused by excessive temperature contact with the patient's surface, and hazards resulting from electrical dangers caused by excessive temperature contact with the patient's surface.
Mechanical Hazards: Including hazards caused by insufficient electric bed support strength, loose or broken accessories, falling of hanging objects, and inadequate angles of moving parts.
Acoustic Hazards: Primarily referring to hazards caused by noise.
4.2 Biological and Chemical Hazards
Hazards resulting from contact with patient and user parts, as well as residues of cleaning agents and disinfectants.
4.3 Operational Hazards
Including hazards caused by malfunctioning controllers, wear and tear of mechanical parts, damage to electrical components, improper installation and operation according to the operating instructions, excessive pulling of control cable, and random placement of the controller after use, as well as the impact of wireless controllers on other electric beds.
4.4 Information Hazards
Missing or incorrect markings, incorrect marking location, inability to be correctly identified, inability to be permanently affixed and clearly visible, non-compliance with regulatory and standard instructions, failure to warn about hazards arising from use with other devices, failure to warn about hazards resulting from long-term use that may lead to functional loss, and failure to warn about foreseeable misuse.
Table 2: Initial Incident and Environmental Examples
| Category | Initial Event and Environmental Examples |
|---|---|
| Incomplete Requirements: |
- Mechanical components not properly assembled; insufficient stroke length of electric actuator; support force requirements not met by bed frame material; bed surface lift stroke not compliant; overall bed angle, bed surface and component angles/folds not compliant; hazards to operators and patients due to improper installation of accessories, inadequate isolation/protection of electrical parts from accessible metal parts, casings, and application parts, insufficient dielectric strength, leading to inadequate protection against electric shock, potentially causing electric shock hazards to users or patients; inadequate liquid ingress protection, leading to hazards; accessories and hanging objects not securely attached, locking mechanisms for wheeled devices not functioning properly; rough surfaces on the device, corners, and edges potentially causing mechanical injury to users or patients; electromagnetic interference exceeding the specified limits, interfering with the normal operation of other equipment; wireless controller interfering with or misoperating other beds. Whether the bed's movement and the folding angles of the boards can be achieved through the controller to meet the specified requirements.
- Biocompatibility issues of materials in contact with the human body.
- The operating manual does not specify the maintenance and care methods, frequency, and disinfection methods for the device and accessories, leading to the device not functioning properly.
- The operating manual does not specify the service life and storage life of the device/accessories, leading to the device/accessories being used beyond their lifespan, resulting in reduced stability and potential safety hazards. Manufacturing Process:
- Unverified changes to control procedures, resulting in device performance parameters not meeting standard requirements.
- Lack of monitoring of critical process control points in the production process, leading to improper component assembly.
- Improper selection of external component suppliers, lack of effective incoming inspection of purchased/outsourced components, resulting in the production of non-conforming purchased/outsourced components, such as: insufficient stroke length of electric actuator, insufficient starting torque, substandard components for head/footboards, and substandard wheel strength. Transportation and Storage:
- Improper product protection leading to damage during transportation.
- Storing the device in an environment exceeding the specified storage conditions (temperature, humidity, pressure), leading to the device not functioning properly. Environmental Factors:
- Excessive temperature or cold may cause the device to malfunction.
- Exposure to strong acids and bases may cause damage.
- Poor electromagnetic interference resistance, leading to malfunction of the device in specific
Table 3: Relationship between hazards, foreseeable events, hazard situations, and potential harm.
| Hazard | Potential Sequence of Events | Hazardous Situation | Harm |
|---|---|---|---|
| Electromagnetic Interference (EMI) | Interference from other equipment in the room causing electrical equipment to start or operate on the bed. | The bed fails to operate according to the controller's instructions. | Patient's condition worsens, death. |
| Movement Components (Inappropriate placement of foot pedal switch on the base) | (1) Accidental stepping; (2) Unlocking of the floor brake locking device. | Bed movement. | Patient's organ damage, worsening condition. |
| Loss or Damage of Function (Wear and tear of moving linkages, manufacturing defects, damage to electric pusher) | (1) Long-term wear and tear of moving parts; (2) Manufacturing defects. | Sudden movement of bed components. | Patient's organ damage, worsening condition, death. |
| Operation (Misuse of Controller) | (1) Improper placement; (2) Accidental contact with function keys. | Unintended movement of the electric bed. | Patient falls, organ damage, worsening condition, death. |
| Incomplete Instructions for Use (Installation of accessories) | (1) Instructions for use do not specify components/accessories; (2) Instructions for use do not specify component installation; (3) Instructions for use do not specify component load capacity; (4) Incorrect component installation. | Components loosen, inability to achieve correct positioning, support structure breaks. | Organ damage, worsening condition, death. |
Tables 2 and 3, based on Appendix E of YY/T 0316-2008 "Application of Risk Management for Medical Devices," provide a suggestive list of initial events and environmental factors that may pose hazards for electric hospital beds. The tables exemplify the relationships between hazards, foreseeable events, hazardous situations, and potential harm, providing guidance and reference for reviewers.
Due to the differences in the principles, functions, and structures of electric beds, the risk factors and examples provided in this chapter are common rather than exhaustive. The above section is only a component of the risk management process, and not the entire risk management process. The applicant for registration should establish, document, and maintain a continuous process in accordance with the procedures and methods specified in YY/T 0316-2008 "Application of Risk Management to Medical Devices for Medical Devices," to identify, assess, and evaluate the risks associated with medical devices, control these risks, and monitor the effectiveness of the controls, in order to ensure the safety and effectiveness of the product throughout its entire lifecycle.
(VIII) The main technical requirements of the product should include:
The main technical indicators of electric beds should include three parts: basic dimensions, performance indicators, and safety indicators. The basic technical indicators listed in this clause are typical indicators for electric beds and accessories. The applicant for registration should refer to relevant national standards, industry standards, and make specific provisions for each indicator based on clinical needs, the technical characteristics of their own products.
- Basic dimensions
Bed: Bed length, bed width (outer edge), overall height (lowest position, with or without mattress).
Mattress: Mattress length, mattress width.
Accessories: Key geometric dimensions (length, width, height, diameter, angle) of accessories.
- Performance Requirements
Bed:
2.1 Typical operating parameters include bed lift stroke, bed longitudinal tilt, bed lateral tilt, backrest angle, legboard angle, footboard angle, and bed longitudinal translation.
2.2 Maximum safe load.
2.3 Smoothness of movement.
2.4 Bed movement and locking.
2.5 X-ray transmissivity (if applicable).
2.6 Appearance.
2.7 Leakage of hydraulic and/or pneumatic systems (if applicable).
Accessories: Specific requirements should be made based on the specific situation of different accessories, such as the adjustment range of the infusion rack.
- Environmental Conditions
After conducting tests according to GB/T 14710-2009, all performance characteristics of the electric bed must meet the requirements.
- Safety Requirements
The safety requirements for electric hospital beds should meet the requirements of GB 9706.1-2007 "Medical Electrical Equipment Part 1: General Safety Requirements," YY 0505-2012 "Medical Electrical Equipment Part 1-2: General Safety Requirements - Parallel Standard: Electromagnetic Compatibility - Requirements and Test Methods," and YY 0571-2013 "Medical Electrical Equipment Part 2: Specific Safety Requirements for Hospital Electric Beds."
(9) Principles and Examples for Determining Typical Products within a Single Registration Unit
The typical product should be the product within the same registration unit that best represents the safety and effectiveness of other products within that unit.
The product with the most complete functionality, the most complex structure, and the highest risk should be considered.
If the main safety and performance indicators of various products within a registration unit are not fully covered by a single product, the product with the most comprehensive coverage of safety and performance indicators should be selected as the typical product. It should also be considered whether other products have safety and performance indicators not covered by the typical product.
For example, if two models within the same registration unit have identical product structures but differ only in X-ray transmission requirements, the model that can transmit X-rays through the material should be selected as the typical product.
If there is insufficient evidence to demonstrate that the electromagnetic compatibility performance of different models within the same registration unit can be covered, each model should be subjected to electromagnetic compatibility testing.
(Ten) Product Manufacturing Requirements
During the manufacturing of an electric bed, the selection and procurement of raw materials directly affect the product's performance. The applicant should carefully review the qualifications of qualified suppliers. During the procurement inspection, the material specifications should be carefully reviewed to ensure compliance with the procurement requirements. The welding and assembly process of the bed frame should be carefully inspected, with particular attention paid to the aspects that affect the bed's performance (such as welding quality and stability).
The applicant shall, based on the actual production process of the product being declared, provide a detailed description of the production process in the form of a flowchart, and describe each process control point in the flowchart. For critical operations and special processes, detailed descriptions of the production operations, methods, and precautions shall be provided in the operating instructions. The operating instructions shall clearly avoid ambiguity for the production personnel. The inspection of critical operations shall be carried out by qualified inspection personnel with relevant training and experience. The following is an example of the production process flowchart for electric hospital beds. The applicant shall develop a suitable production process flowchart for their enterprise based on the actual production situation:
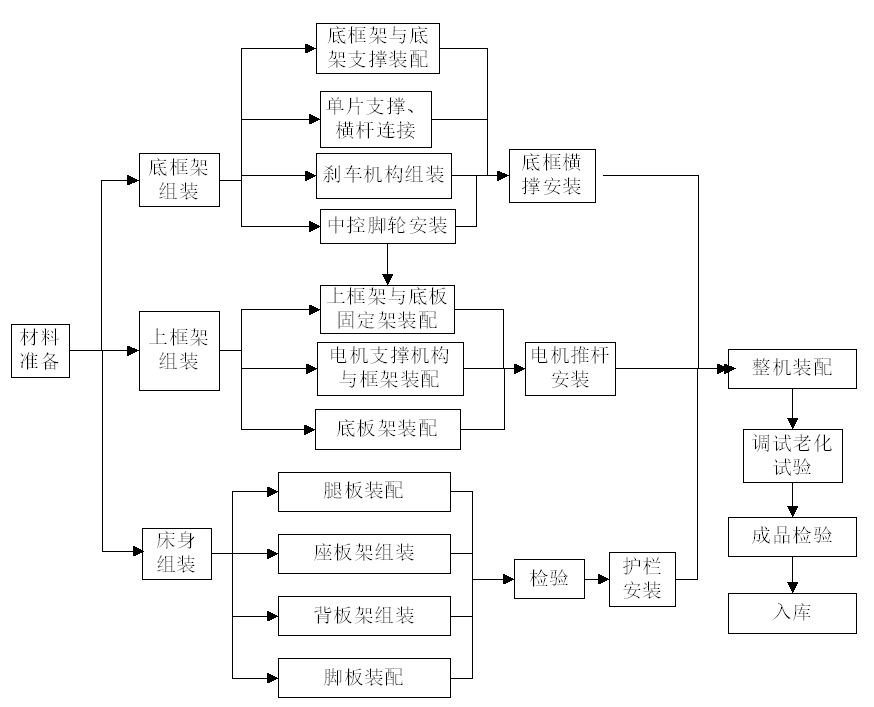
Figure 3: Example of an electric hospital bed manufacturing process flow diagram.
(11) Clinical Evaluation Requirements for the Product
The applicant shall submit clinical evaluation data for the electric bed in accordance with the requirements of the "Second Class Medical Device Exemption from Clinical Trials Directory" and the "Technical Guidance on Clinical Evaluation of Medical Devices."
(12) History of Adverse Events for this Type of Product
No relevant reports have been found. The applicant should pay attention to the adverse event history of similar products during risk analysis.
(XIII) Requirements for Product Instructions and Labels
- The instructions for the electric bed should comply with the requirements of the "Regulations on the Management of Medical Device Instructions and Labels" (Order No. 6 of the National Medical Products Administration) and relevant standards. When different models and specifications are sold separately, separate instructions should be provided.
The instructions for the electric bed should also include, at a minimum, the following:
1.1 The operating conditions of the electric bed, particularly information regarding potential electromagnetic interference or other interference between the device and other equipment, as well as recommendations for avoiding such interference.
1.2 Safety Precautions: Clearly state emergency handling procedures for abnormal situations; precautions for specific situations (power outages, accidental movement, etc.); ensure reliable grounding of the power supply; potential for misuse and potential harm resulting from misuse; specify whether the use of other accessories or materials reduces the minimum safety level, and whether these accessories or materials are described; warnings regarding auxiliary power supplies that cannot be maintained in a fully functional state; safe usage period; instructions on parts that should not be directly contacted by patients or users.
1.3 Product Structure: Review whether the product's structure and accessories are clearly defined, and whether the functions of the product and each accessory are clearly defined; Product Performance Specifications: Review whether the product's performance specifications are covered by the product's technical requirements, whether the main performance and parameters are clearly defined, and whether the shape and installation dimensions are clearly defined.
1.4 Installation and Debugging: Review whether the responsible party for product installation and debugging is clearly defined (i.e., whether on-site installation and debugging are provided); whether the installation, debugging methods and precautions for parts that require user self-installation (such as detachable accessories) are clearly defined; maintenance after long-term inoperability.
1.5 Instructions for Use: This document primarily covers pre-use inspection and preparation; operating procedures, methods, and precautions during operation; and shutdown procedures and precautions.
1.6 Fault Analysis and Troubleshooting: This document reviews potential faults and analyzes the causes of these faults; clarifies which faults must be addressed by the manufacturing unit and which must be addressed by the user; and provides methods for the user to resolve the identified faults.
1.7 Maintenance and Servicing: Review whether the instructions clearly specify the methods and frequency for daily maintenance and servicing; maintenance and servicing methods during operation; inspection, maintenance, and replacement cycles and methods for auxiliary power; maintenance and servicing cycles; and clearly defined cleaning methods (e.g., surface wiping, spray cleaning, etc.) and long-term maintenance and servicing methods.
1.8 Explanation of Special Symbols: Review whether the special symbols used near operating and control components and in the vicinity of each component's mounting holes, symbols used on packaging, and symbols specified in GB 9706.1-2007 "Medical Electrical Equipment Part 1: General Safety Requirements" and YY 0571-2013 "Medical Electrical Equipment Part 2: Special Safety Requirements for Hospital Electric Beds" 6.1 are explained.
1.9 Replacement methods for circuit breakers and other components (if applicable).
1.10 Circuit diagrams, component lists: Review whether the necessary information, such as circuit diagrams, component lists, diagrams, and correction details, required to be provided by the manufacturer for parts that users need to repair themselves, is clearly specified.
1.11 Cleaning methods before use: The manufacturer should list these according to the specific product.
1.12 Product structure diagrams, photos, attachments, and a detailed list of specialized tools, etc.
1.13 Provide a statement confirming compliance with electromagnetic compatibility requirements, as specified in YY0505-2012.
1.14 Software release version.
- Labeling
The product label must comply with the requirements of "Regulations on the Management of Instructions and Labels for Medical Devices" (State Administration for Market Regulation, Decree No. 6) and YY/T 0466.1-2009 "Symbols for Labels, Marking and Information Provision on Medical Devices", etc.
(Fourteen) Product Research Requirements
- Performance Research Data
1.1 Verification of performance criteria should be based on the product structure composition as described in the relevant documentation, and may refer to the requirements of YY 0571-2013 "Medical Electrical Equipment Part 2: Special Safety Requirements for Hospital Electric Beds." This should provide the basis for determining functional and safety indicators (such as electrical safety and electromagnetic compatibility, radiation safety), as well as other indicators related to quality control, including the standard(s) or method(s) used, the reasons for their use, and the underlying theory. At a minimum, the following indicators should be included: bed body lifting stroke, longitudinal tilt of the bed body, lateral tilt of the bed body, backrest folding angle, legboard folding angle, footboard folding angle, longitudinal displacement of the bed surface, maximum safe load, smoothness of operation, movement and locking of the bed, X-ray transmissivity (if applicable), appearance, noise, etc. If accessories are included, corresponding requirements should be made based on the specific situation of each accessory, such as the adjustment range of the infusion stand.
1.2 Verification of Safety Indicators includes two main categories: electrical safety indicators and electromagnetic compatibility indicators. Electrical safety indicators should include all indicators from GB 9706.1-2007 "Medical Electrical Equipment Part 1: General Safety Requirements" and YY 0571-2013 "Medical Electrical Equipment Part 2: Specific Safety Requirements for Electric Beds in Hospitals" and other applicable national and industry standards. Electromagnetic compatibility indicators should include all indicators from YY 0505-2012 "Medical Electrical Equipment Part 1-2: General Safety Requirements - Parallel Standard: Electromagnetic Compatibility - Requirements and Tests" and other applicable national and industry standards.
The research materials should clearly specify the structural components, main component information, production bed frame, headboard, and footboard material names, brand, specifications, and other details determined through the research verification.
- Biological Compatibility Evaluation
The biological compatibility evaluation should be conducted in accordance with the requirements of GB/T16886.1-2011 "Medical Device Biological Evaluation Part 1: Evaluation and Tests in the Risk Management Process" based on the contact site, contact method, and contact time of the product with the human body.
Disinfection Method Research: The applicant should specify the recommended disinfection process (method and parameters) and the basis for the recommended disinfection method, based on the materials used in the manufactured product.
Product Shelf Life and Packaging Research
Shelf Life Research: A verification report demonstrating the product's shelf life must be provided. The report should provide a detailed description of the consumable, fragile, or components requiring periodic replacement (such as casters, brakes, and batteries) included in the declared product, as well as the specific reasons for determining the product's shelf life or expiration date.
Packaging Research: The packaging materials for the product must be clearly specified. Verification data demonstrating compliance with transportation test requirements under the declared transportation conditions must be provided. Furthermore, the basis for maintaining the integrity of the packaging under the declared storage conditions must be provided.
- Software Research (if applicable)
Refer to the relevant requirements in the "Guidance Principles for Registration of Medical Device Software".
5.1 This product is a product containing embedded software, and a separate embedded software description document should be provided. Based on the potential risks to patients that the product's intended use may cause, determine the safety level of the software being declared, and submit a random software description document according to the determined safety level.
5.2 Provide a statement regarding the naming rules for software versions, clearly specifying all fields and the meaning of each field in the software version, and determining the complete version and the identifier version used for release. Specifically, the complete version information of the software should be consistent with the corresponding content in the random software description document, and the identifier version information used for release should be consistent with the content of the product manual and the random software description document.
Technical Guidance on Registration of Electric Hospital Beds
I. Purpose and Background of Writing the Guidance
(One) The purpose of this guideline is to guide and standardize the technical review work for electric hospital beds. It aims to help reviewers understand and master the principles/mechanisms, structure, performance, and intended use of this type of product, as well as to grasp the basic requirements and standards for technical review, and to conduct a systematic evaluation of the product's safety and effectiveness.
(Two) With the promulgation and implementation of the "Medical Device Supervision and Administration Regulations" (No. 650 of the State Council of the People's Republic of China) and the issuance of the "Provisional Management Regulations for Registration of Medical Devices" (No. 4 of the State Administration for Market Regulation of Medicines and Food), the original technical guidance principles for electric hospital beds, which were previously established, no longer meet the current regulatory requirements. This guideline is revised in accordance with the new regulations to meet the current regulatory requirements.
II. Basis for Guideline Development
(One) "Medical Device Supervision and Administration Regulations" (No. 650 of the State Council of the People's Republic of China)
(Two) "Provisional Management Regulations for Registration of Medical Devices" (No. 4 of the State Administration for Market Regulation of Medicines and Food)
(Three) "Regulations on the Management of Medical Device Instructions and Labels" (No. 6 of the State Administration for Market Regulation of Medicines and Food)
(Four) "Announcement Regarding the Requirements for Registration Declaration Materials and the Format of Approval Documents for Medical Devices" (National Medical Products Administration Announcement No. 43 of 2014)
(Five) "Announcement Regarding the Publication of the List of Second-Class Medical Devices Exempt from Clinical Trials" (National Medical Products Administration Announcement No. 12 of 2014)
(Six) "Announcement Regarding the Publication of Technical Requirements Writing Guidelines for Medical Devices" (National Medical Products Administration Announcement No. 9 of 2014)
(Seven) "Technical Guidance Principles for Clinical Evaluation of Medical Devices" (National Medical Products Administration Announcement No. 14 of 2015)
(Eight) "Technical Review Guidance Principles for Medical Device Software Registration" (National Medical Products Administration Announcement No. 50 of 2015)
(Nine) Notice Regarding the Issuance of the "Guidance on Biological Evaluation and Review of Medical Devices" (National Food and Drug Administration Device [2007] No. 345)
(Ten) GB 9706.1-2007 "Medical Electrical Equipment Part 1: General Safety Requirements"
(Eleven) YY 0571-2013 "Medical Electrical Equipment Part 2: Special Safety Requirements for Hospital Electric Beds"
(12) YY 0505-2012 "Medical Electrical Equipment, Part 1-2: Safety General Requirements and Parallel Standards, EMC Requirements and Tests."
III. Key Content Explanation
(1) In the requirements for product names, it refers to the "Medical Device Classification Directory" and YY 0571-2013 "Medical Electrical Equipment, Part 2: Specific Safety Requirements for Hospital Electric Beds," as well as other relevant requirements, thereby standardizing the principles of product naming.
(2) In the structural and composition content, a typical electric bed's structural diagram is provided, and a brief introduction to the main structure and accessories of electric beds is given.
(3) In the product's working principle, the working principle of electric beds is simply introduced.
(4) Since the product is a non-therapeutic medical device, this guideline does not include the product's mechanism of action.
(5) The relevant standards applicable to the product include the current and effective national standards, industry standards (including product standards, basic standards).
(Six) Regarding the main risks of the product, the following requirements were established based on YY/T 0316-2008 "Application of Risk Management for Medical Devices" and its appendices C, E, F, G, I, and J, concerning the safety characteristics, hazards, foreseeable event sequences, and hazard situations of electric beds, as well as the risk control scheme and implementation, and the overall evaluation of residual risks and relevant methods for production and post-production monitoring. Furthermore, a systematic analysis of the hazards, foreseeable event sequences, and hazard situations of electric beds was conducted.
(Seven) The main performance indicators of the product include all aspects that need to be considered, with some requiring reference to relevant national standards and industry standards, while others require consideration based on the technical capabilities of the enterprise.
(Eight) In the product's instruction manual, the key areas for review of the electric bed were specified, such as performance, precautions, use, maintenance, and storage.
Four. Compilation Unit
Tianjin Medical Device Technology Appraisal Center.

